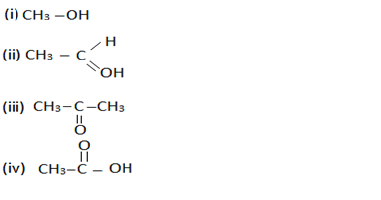Class 10 Science questions of a set of important 50 questions for-2020-21 CBSE Board.
Class 10 Science questions of a set of important 50 questions for-2020-21 CBSE Board will help you in preparations of science subject of your CBSE board exam. The problems in the set of 50 science questions are selected from last year’s question papers and sample papers. All questions are written beautifully by a subject expert of future study points, so we hope each student will understand what type of questions are asked in the CBSE Board exam.
The importance of studying a set of 50 important science questions is mandatory for each student since this set of questions is selected as per the latest CBSE curriculum. It is important for every class 10 CBSE student to get the idea of the type of science questions asked in the CBSE board exam. Each student would be able to know to get the idea of questions they are going to see in the science question paper of 2020-21 board exam.
Click for online shopping
Future Study Point.Deal: Cloths, Laptops, Computers, Mobiles, Shoes etc
Here in future study point, you can study science and maths solutions of NCERT syllabus, sample papers, and the solutions of previous year’s question paper. We take care of our students,it is that’s why we also write carrier-related blogs which help the students in building their carrier also.
Solutions : Half yearly maths paper of class 10 CBSE 2020
NCERT Solutions Class 10 Science from chapter 1 to 16
NCERT Solutions of all chapters of Maths for Class 10 from Chapters 1 to 15
Type of Chemical Reactions with Complete detail(Notes)
What is pH value and its importance in everyday life.(Notes)
Anatomy of the Humane brain-Class 10 CBSE(Notes)
Special notes-Refraction,refraction,dispersion and scattering of light(Notes)
Special notes-Structure and function
Special note-Myopia, Hypermetropia and Presbyopia
Complete detail of electrical resistance and conductance(Notes)
Electric Current and Heating effect of Electric Current(Notes)
Magnetic effect of electric current
Ozone Layer and How it is Getting depleted(Note)
Science and Maths NCERT solutions for Class 9 ,10 and 11 classes
Q1.List two causes of presbyopia. Draw a labeled diagram of a lens used for the correction of this defect of vision.
Q2.Show the formation of Na2O by the transfer of electrons.
Q3.List in tabular form two differences between pepsin and trypsin.
Q4.Name the process used to harness nuclear energy these days. Explain it briefly. List two advantages of using nuclear energy in place of fossil fuels.
Q5.Why are forests considered ‘‘biodiversity hot spots’’ ? Suggest four approaches towards the conservation of forests.
Q6.What is atmospheric refraction? Explain with the help of a labeled diagram that the position of a star as seen by us is not its true position.
Q7.Write the chemical formula of washing soda. How can it be obtained from baking soda? List two industries in which washing soda is used for other purposes than washing clothes
Q8.Out of three metals P, Q, and R, P is less reactive than Q and R is more reactive than P and Q both. Suggest an activity to arrange P, Q, and R in order of their decreasing reactivity.
Q9.(a) How is the possible valency of an element determined from the electronic configuration of its atom?
(b) Determine the valency of an element X whose atomic number is 15.
Q10.What is hemoglobin? State the consequences of deficiency of hemoglobin in our bodies.
Q11.Explain the following :
(a) Speciation
(b) Natural Selection
Q12.Explain the ways in which glucose is broken down in the absence or shortage of oxygen.
Q13.List in tabular form three distinguishing features between cerebrum and cerebellum.
Q14.(a) What is a hydrocarbon? Give its one example.
(b) Give the structural difference between saturated and unsaturated hydrocarbons with two examples each.
(c) Name the following compounds.
Q15.(a) What is a double displacement reaction? Explain with an example.
(b) A small amount of quick lime is added to water in a beaker.
(i) Name and define the type of reaction that has taken place.
(ii) Write the balanced chemical equation for the above reaction and the chemical name of the product formed.
(iii) List two main observations of this reaction.
Answers of most important science questions 2020-21 CBSE Board
Q16.(a) List four characteristics of the image formed by a concave lens of focal length 20 cm when the object is placed at a distance of 40 cm from its optical center.
(b) The size of an image of an object by a convex lens of focal length 20 cm is observed to be reduced to 3 1 rd of its size. Find the distance of the object from the optical center of the lens.
Q17. Three resistors of resistances R1 , R2, and R3 are connected (i) in series, and (ii) in parallel. Write expressions for the equivalent resistance of the combination in each case.
(b) Two identical resistors of 12 Ω each are connected to a battery of 3 V. Calculate the ratio of the power consumed by the resulting combinations with minimum resistance and maximum resistance.
Q18. (a) Name and state the rule to determine the direction of force experienced by a current-carrying straight conductor placed in a uniform magnetic field that is perpendicular to it.
(b) Draw a labeled diagram of an electric motor.
Latest electronic items,mobiles,laptops and desktops at easy instalments
Q19. How is O2 and CO2 transported in human beings?
Q20.Write the structure of the eye lens and state the role of ciliary muscles in the human eyes.
Q21. 2 g of silver chloride in a china dish and the china dish is placed in sunlight for some time. What will be your observation in this case? Write the chemical reaction involved in the form of a balanced chemical equation. Identify the type of chemical reaction.
Q22. Identify the type of chemical reaction taking place in each of the following cases and write the balanced chemical equation for the reactions.
(a) Zinc reacts with silver nitrate to produce zinc nitrate and silver.
(b) Potassium iodide reacts with lead nitrate to produce potassium nitrate and lead oxide.
Q23. List two differences between acquired traits and inherited traits by giving one example of each.
Q24.Why should there be an equitable distribution of resources? List three forces that would be working against an equitable distribution of our resources.
Q25.How can we help in reducing the problem of waste disposal? Suggest any three methods.
10 th Class previous years question papers and sample papers of maths and science
Q26. Define an ecosystem. Draw a block diagram to show the flow of energy in an ecosystem.
Q27. Write the chemical formula and name of the compound which is the active ingredient of all alcoholic drinks. List its two uses. Write chemical equations and name of the products formed when this compound reacts with-
(i) Sodium metal
(ii) Hot concentrated sulphuric acid
Q28. What is methane? Draw its electronic dot structure. Name the type of bonds formed in this compound. Why are such compounds?
(i) Poor conductor of electricity and
(ii) Have low melting and boiling points? What happens when these compounds burn in oxygen?
Q29(a)Write chemical equations for the following reactions:
(i) calcium metal reacts with water.
(ii)Cinaabar is heated in the presence of air.
(iii)manganese dioxide is heated with aluminum powder.
(b).What are alloys? List two properties of alloys.
CBSE Class 10 science question paper 2020 SET -3 solutions
Q30.An object is placed at a distance of 30cm from a concave lens of focal length 30cm.
(i)Use the lens formula to determine the distance of the image from the lens.
(ii)List four characteristics of the image (nature position, size, erect/inverted)in this case.
(iii)Draw a labeled diagram to justify your answer to part(ii).
Q31(a)With the help of a suitable circuit diagram proves that the
reciprocal of the equivalent resistance of a group of resistances joined in parallel is equal to the sum of the reciprocals of the individual resistances.
(b)I an electric circuit two resistors of 12Ω each are joined in parallel to a 6V battery. Find the current drawn from the battery.
Q32. An electric lamp of resistance 20Ω and a conductor of resistance 4Ω are connected to a 6V battery as shown in the circuit. Calculate:
(a)The total resistance of the circuit.
(b)The current through the circuit.
(c)The potential difference across the (i)electric lamp and (ii) conductor, and
(d)Power of the lamp
Q33.What is a solenoid? Draw the pattern of magnetic field lines
(i)A current-carrying solenoid and (ii) a bar magnet.List two distinguishing features between the two fields.
Q33.Define pollination. Explain the different types of pollination lead to fertilization?
In the reaction :
(a) Name the compound (i) oxidized, (ii) reduced.
(b) Define oxidation and reduction on its basis.
Q34.1 g of solid sodium chloride is taken in a clean and dry test tube and 2 mL of conc. sulphuric acid is added to it. If the gas evolved is tested first with dry and then with wet blue litmus paper, in which case will the litmus paper change colour ? Give the reason for your answer. What inference can be drawn about the nature of the evolved gas? Support your answer with the chemical equation for the reaction.
Q35.(a) For the preparation of cakes, baking powder is used. If at home your mother uses baking soda instead of baking powder, how will it affect the taste of the cake and why?
(b) How is baking soda be converted into baking powder?
(c) What makes the cake soft and spongy?
Q37.What is geotropism? Draw a labeled diagram of a potted plant showing positive geotropism and negative geotropism.
Q38.We wish to obtain an equal-sized inverted image of a candle flame on a screen kept at a distance of 4 m from the candle flame.
(a) Name the type of lens that should be used.
(b) What should be the focal length of the lens and at what distance from the candle flame the lens be placed.
(c) Draw a labeled diagram to show the image formation in this case.
Q39.Calculate the total cost of running the following electrical devices in the month of September, if the rate of 1 unit of electricity is Rs 6.00.
(i) Electric heater of 1000 W for 5 hours daily.
(ii) Electric refrigerator of 400 W for 10 hours daily.
Q40.(a) What is the variation? How is a variation created in a population? How does the creation of variation in a species promote survival?
(b) Explain how, offspring and parents of organisms reproducing sexually have the same number of chromosomes.
Solutions of maths question paper 2020 CBSE board set 1
Q41.(a) What are magnetic field lines? How is the direction of the magnetic field at a point in a magnetic field determined using field lines?
(b) Two circular coils ‘X’ and ‘Y’ are placed close to each other. If the current in the coil ‘X’ is changed, will some current be induced in the coil ‘Y’? Give reason.
(c) State ‘Fleming’s right-hand rule”.
Q42. (b) Write the mechanism by which fishes breathe in water.
(c) Name the balloon likes structures present in the lungs. List its two functions.
(d) Name the respiratory pigment and write its role in human beings.
Q43.(a) Name the process and explain the type of nutrition found in green plants. List the raw materials required for this process. Give the chemical equation for the mentioned process.
(b) Write three events that occur during this process.
Q44. Explain giving justification the trends in the following properties of elements, on moving from left to right in a period, in the Modern Periodic Table.
(a) Variation of valency.
(b) Change of atomic radius.
(c) Metallic to the non-metallic character.
(d) Electronegative character.
(e) Nature of oxides.
Q45. (a) List in the tabular form any three chemical properties on the basis of which metals and non-metals are differentiated.
(b) State two ways to prevent the rusting of iron.
Q46. What is “Sustainable Management of Natural Resources” ? Why is it necessary ? Which one out of reuse and recycle, would you practice in your daily life and why?
Q47.While teaching the chapter “Our Environment” the teacher stressed the harmful effects of burning of fossil fuels, plastic, paper, etc. The students noticed the extensive use of plastic and polythene in daily life, which can be avoided and the surroundings can be kept clean. They decided to make their school “plastic and polythene” free and motivated each other for its minimum use.
(a) Why should the use of polythene and plastic be reduced in daily life?
(b) In what ways the students would have avoided the use of plastic and polythene in their school?
(c) How the students would have motivated each other for the success of their decision?
Q48.Calculate the total cost of running the following electrical devices in the month of September, if the rate of 1 unit of electricity is ` 6.00.
(i) Electric heater of 1000 W for 5 hours daily.
(ii) Electric refrigerator of 400 W for 10 hours daily.
Q49.What is electrical resistivity? Derive its SI unit. In a series electrical circuit comprising a resistor made up of a metallic wire, the ammeter reads 100 mA. If the length of the wire is doubled, how will the current in the circuit change ? Justify your
answer.
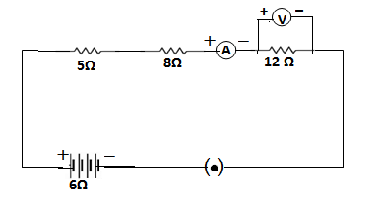
Q50.Consider the following circuit :
What would be the readings of the ammeter and the voltameter when the key is closed? Give reasons to justify your answer.
Q51.Calculate the total cost of running the following electrical devices in the month of September, if the rate of unit of electricity is ₹6.00.
(i)Electric heater of 1000W for 5 hours daily.
(ii)Electric refrigerator of 400W for 10 hours daily.
Study notes of Maths and Science NCERT and CBSE from class 9 to 12
Government jobs after 10 or 12 th pass : Qualify Entrance exams of SSC MTS and SSC CHSL
Tips to get success in competitive exams
You can compensate us by donating any amount of money for our survival
Our Paytm NO 9891436286
NCERT Solutions of Science and Maths for Class 9,10,11 and 12
NCERT Solutions for class 9 maths
NCERT Solutions for class 9 science
NCERT Solutions for class 10 maths
CBSE Class 10-Question paper of maths 2021 with solutions
CBSE Class 10-Half yearly question paper of maths 2020 with solutions
CBSE Class 10 -Question paper of maths 2020 with solutions
CBSE Class 10-Question paper of maths 2019 with solutions
NCERT Solutions for Class 10 Science
NCERT Solutions for class 11 maths
| Chapter 1-Sets | Chapter 9-Sequences and Series |
| Chapter 2- Relations and functions | Chapter 10- Straight Lines |
| Chapter 3- Trigonometry | Chapter 11-Conic Sections |
| Chapter 4-Principle of mathematical induction | Chapter 12-Introduction to three Dimensional Geometry |
| Chapter 5-Complex numbers | Chapter 13- Limits and Derivatives |
| Chapter 6- Linear Inequalities | Chapter 14-Mathematical Reasoning |
| Chapter 7- Permutations and Combinations | Chapter 15- Statistics |
| Chapter 8- Binomial Theorem | Chapter 16- Probability |
CBSE Class 11-Question paper of maths 2015
CBSE Class 11 – Second unit test of maths 2021 with solutions
NCERT solutions for class 12 maths
| Chapter 1-Relations and Functions | Chapter 9-Differential Equations |
| Chapter 2-Inverse Trigonometric Functions | Chapter 10-Vector Algebra |
| Chapter 3-Matrices | Chapter 11 – Three Dimensional Geometry |
| Chapter 4-Determinants | Chapter 12-Linear Programming |
| Chapter 5- Continuity and Differentiability | Chapter 13-Probability |
| Chapter 6- Application of Derivation | CBSE Class 12- Question paper of maths 2021 with solutions |
| Chapter 7- Integrals | |
| Chapter 8-Application of Integrals |
Class 12 Solutions of Maths Latest Sample Paper Published by CBSE for 2021-22 Term 2
Class 12 Maths Important Questions-Application of Integrals
Solutions of Class 12 Maths Question Paper of Preboard -2 Exam Term-2 CBSE Board 2021-22
Solutions of class 12 maths question paper 2021 preboard exam CBSE Solution